Finally, the Control
You Need to Deliver
Accurate Treatment
When you reach for a balloon expandable stent, you require accuracy. The LifeStream® Balloon Expandable Covered Stent was developed using Bard's vast experience in PTA and covered stents to create a device designed for the challenging anatomy of iliac arteries and engineered to facilitate accurate placement. With a design that facilitates ease of trackability, low sheath profile, stent-specific marker bands, and minimal foreshortening, the LifeStream® Covered Stent helps you deliver accurate performance.
Low Sheath Profile
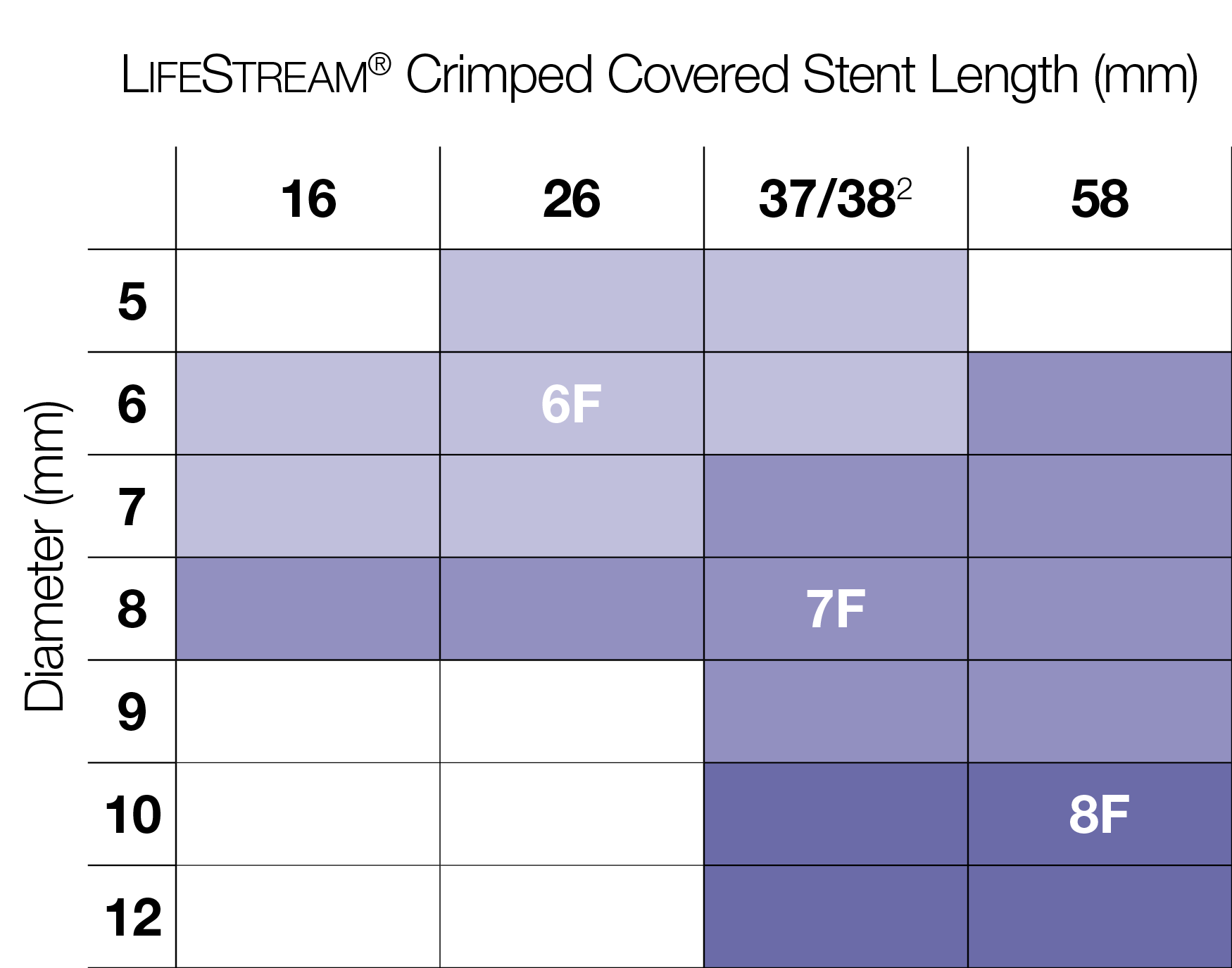
LifeStream® Covered Stent offers sizes on a 6F platform, which is the lowest sheath profile among balloon expandable covered stents on the U.S. market with an iliac indication.1
6F platform for
balloon
expandable
covered stent
Broad Size Matrix
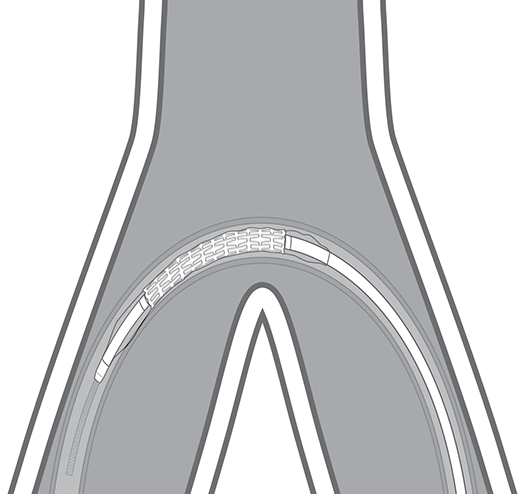
Ease of Delivery
The LifeStream® Covered Stent is designed to provide trackability to reach lesions through complex and tortuous anatomy - providing ease of delivery to the target lesion.
Non-Compliant Balloon Technology
Utilizing non-compliant balloon technology, the LifeStream® Covered Stent is designed to provide precise diameters and efface heavily-calcified iliac lesions.

Designed for Trackability
Accurate Placement
The radiopaque marker bands of the LifeStream® Covered Stent have been specifically positioned on the balloon catheter at the ends of the crimped covered stent to facilitate accurate stent placement. And when millimeters count, the LifeStream® Covered Stent, with an average of 3.5% foreshortening across all balloon sizes at nominal inflation pressure4, achieved a high Acute Technical Success Rate of 98.3% in the BOLSTER Study.5
Stent-Specific Marker Bands
Maximum distance between inner border of marker band and covered stent end on each side = 0.80 mm
Minimal Foreshortening
(7 x 37 mm example)

Optimized for Iliac Interventions
An optimized balloon design with short balloon shoulders and cones helps minimize dilatation of healthy tissue and reduce the risk of catheter entanglement during kissing stent procedures.

BOLSTER Clinical Study
BallOon ExpandabLe Vascular Covered Stent in the Treatment of Iliac ArtERy Occlusive Disease
Study Design
- Device- and/or procedure-related death or MI through 30 days; or
- Any TLR, major limb amputation, or restenosis (DUS) through 9 months
- Technical Success
- Procedure Success
- TLR/TVR
- Primary Patency
The clinical study results demonstrated the safety and effectiveness of the LifeStream® Balloon Expandable Vascular Covered Stent for its intended use. As analyzed on a Pre-Specified basis, the primary composite endpoint result was 16.2% (p-value 0.1987). As analyzed on a Post-Hoc basis utilizing 12-month assessments and additional clinical factors, the primary composite endpoint result was 11.6%.
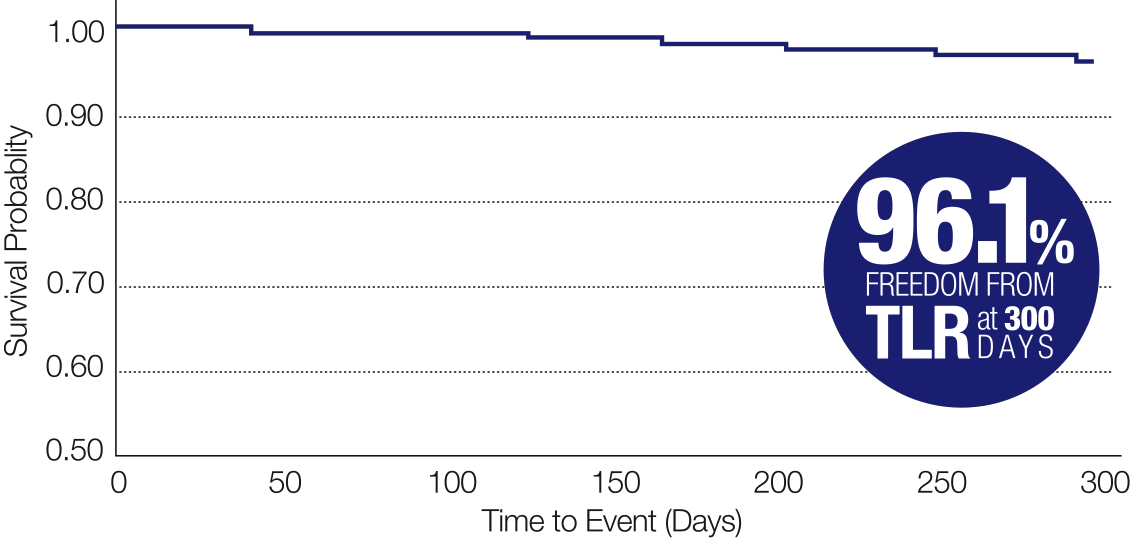
Freedom from TLR6